清華加速器
清華已累積超過100家新創公司的陪跑經驗,以及協助新創從種子期開始一路茁壯至IPO的成功案例; 在清華加速器,我們依據新創團隊所在的不同領域、階段、商業模式,由產業專家群與業師群一對一量身打造加速方案。同時,清華透過面向全球建立創新高速公路 (Innovation Highways),提供新創公司、企業、學研團隊在人才、資本、技術、核心設施、產業鏈結等多維度客制化生態系服務。在對於本土新創與企業的協助上,清華亦透過創新高速公路 (Innovation Highways),鏈結全球關鍵學研、企業夥伴、市場、投資人,協助本土新創與企業面向利基市場拓展產品與服務。
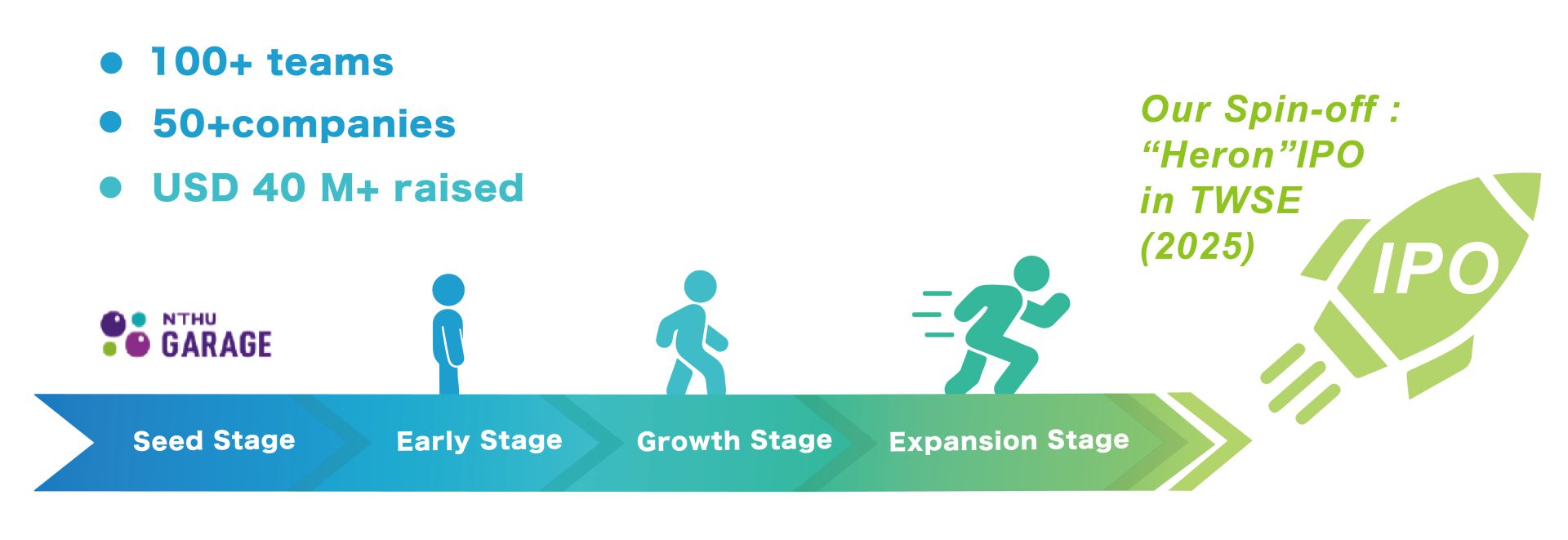
逾十年育成經驗,累計輔導百家以上新創團隊
依新創領域、階段與商業模式量身打造的一對一加速方案
覆蓋從種子期到成長期,並以長期發展與上市潛力為目標
提供場域空間、創業課程與整合型商業諮詢服務
串聯人才、技術、客戶、資本、供應鏈及政府創業資源
Innovation Highways為全球生態系介接台灣資源提供客製化服務
BNCT商化之路 — 從Lab到IPO
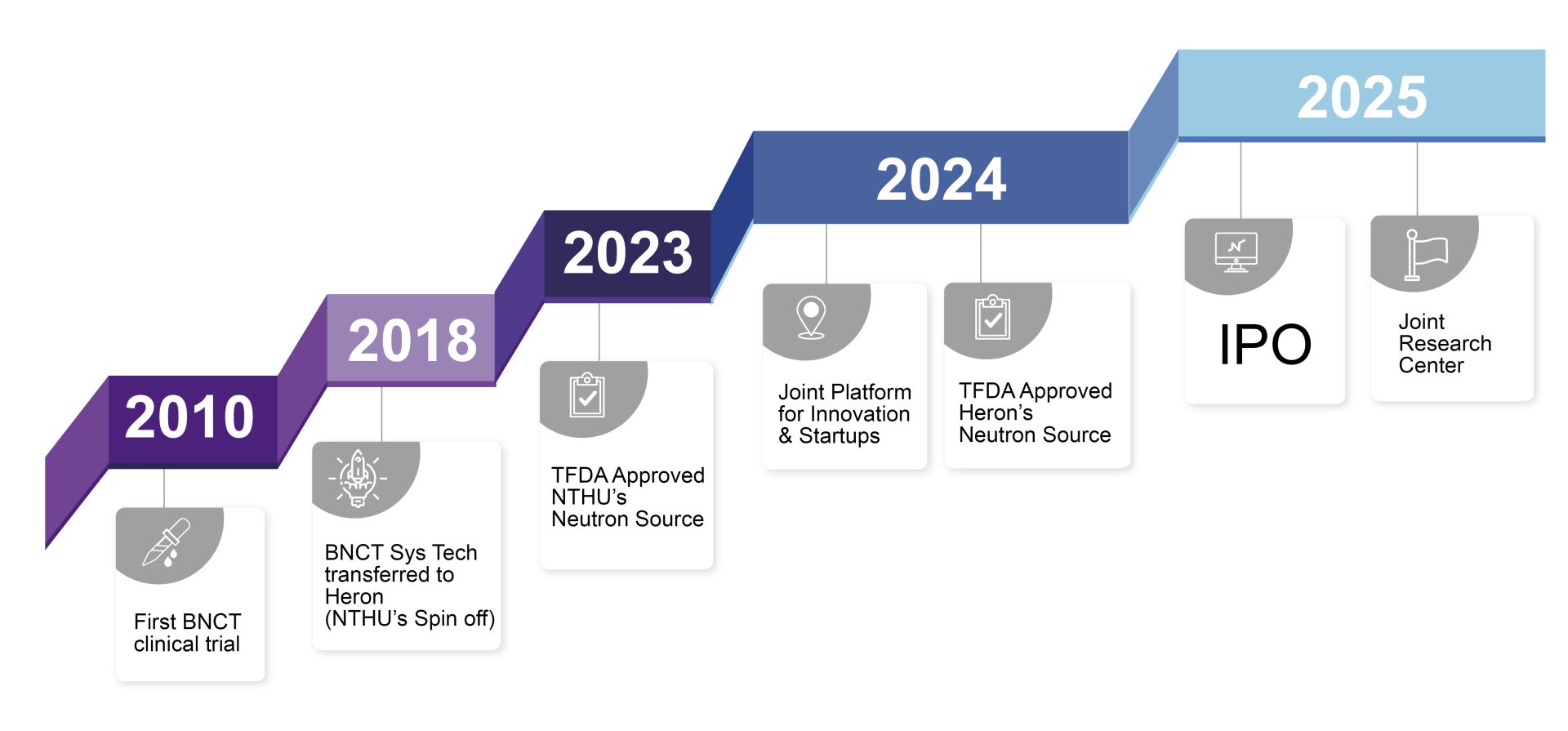
硼中子捕獲治療 (Boron Neutron Capture Therapy,簡稱BNCT) 為下世代的腫瘤治療技術,清華團隊經過多年來在清華中子源累積的 BNCT 研究與臨床基礎,於2018年與工研院、漢民科技合作,將BNCT軟硬體技術移轉至衍生新創- 禾榮公司。由禾榮專注於 BNCT 系統的小型化,以及硬體、軟體和藥物的整合開發。
在產、學的長期合作陪跑下,由清華大學提供IP、技術、科研人才、關鍵設施、育成服務;漢民科技作為禾榮早期主要投資者,提供發展所需資本、市場渠道、運營管理人才,這種互補模式使得清華的反應堆中子源 (Reactor-based neutron source) 順利轉型為適合醫院部署的加速器型中子源 (Accelerator-based neutron source),並於醫院內實現佈署、應用於人體臨床。
在雙邊緊密合作的過程中,清華大學的中子源於2023年首先獲得我國衛服部 (TFDA)之醫療器械認證,隨後禾榮的加速器型中子源亦在 2024 年獲得 TFDA 批准;禾榮公司更於2025年於台灣證交所上市,市值超過千億新台幣。2025年9月,禾榮與清華攜手在清華成立校級聯合研發中心,進一步深化在雙邊 BNCT 藥物、器械等前瞻領域的長期合作。